AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Period table definition chemistry2/26/2024  
From the table above, a single bond between fluorine and nitrogen has a bond length of approximately 64 + 71 =135 pm. The bond between fluorine and nitrogen is a single bond. To find the nitrogen-to-fluorine bond length in NF 3, draw the Lewis structure. Therefore, the bond length is greater in CO 2.ĥ. In general, any element which corresponds to the d-block of the modern periodic table (which consists of groups 3-12) is considered to be a. Therefore, the bond length is greater in CO 2.Īnother method makes use of the fact that the more electron bonds between the atoms, the tighter the electrons are pulling the atoms together. IUPAC defines transition elements as an element having a d subshell that is partially filled with electrons, or an element that has the ability to form stable cations with an incompletely filled d orbital. Referring to the table above, a double bond between carbon and oxygen has a bond length of approximately 67 + 57 = 124 pm and a triple bond between carbon and oxygen has a bond length of approximately 60 + 53 =113 pm. From the Lewis structures for CO 2 and CO, there is a double bond between the carbon and oxygen in CO 2 and a triple bond between the carbon and oxygen in CO. The bond between carbon and nitrogen is a triple bond, and a triple bond between carbon and nitrogen has a bond length of approximately 60 + 54 =114 pm.Ĥ. To find the carbon-nitrogen bond length in HCN, draw the Lewis structure of HCN. Adding these together and dividing by the number of bonds (3) reveals that the bond order of nitrate is 1.33.ģ. N=O has a bond order of two, and both N-O bonds have a bond order of one. To find the bond order of this molecule, take the average of the bond orders. The Lewis structure for NO 3 - is given below: There is a double bond between the two oxygen atoms therefore, the bond order of the molecule is 2.Ģ. First, write the Lewis structure for \(O_2\). It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used.\)ġ. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. 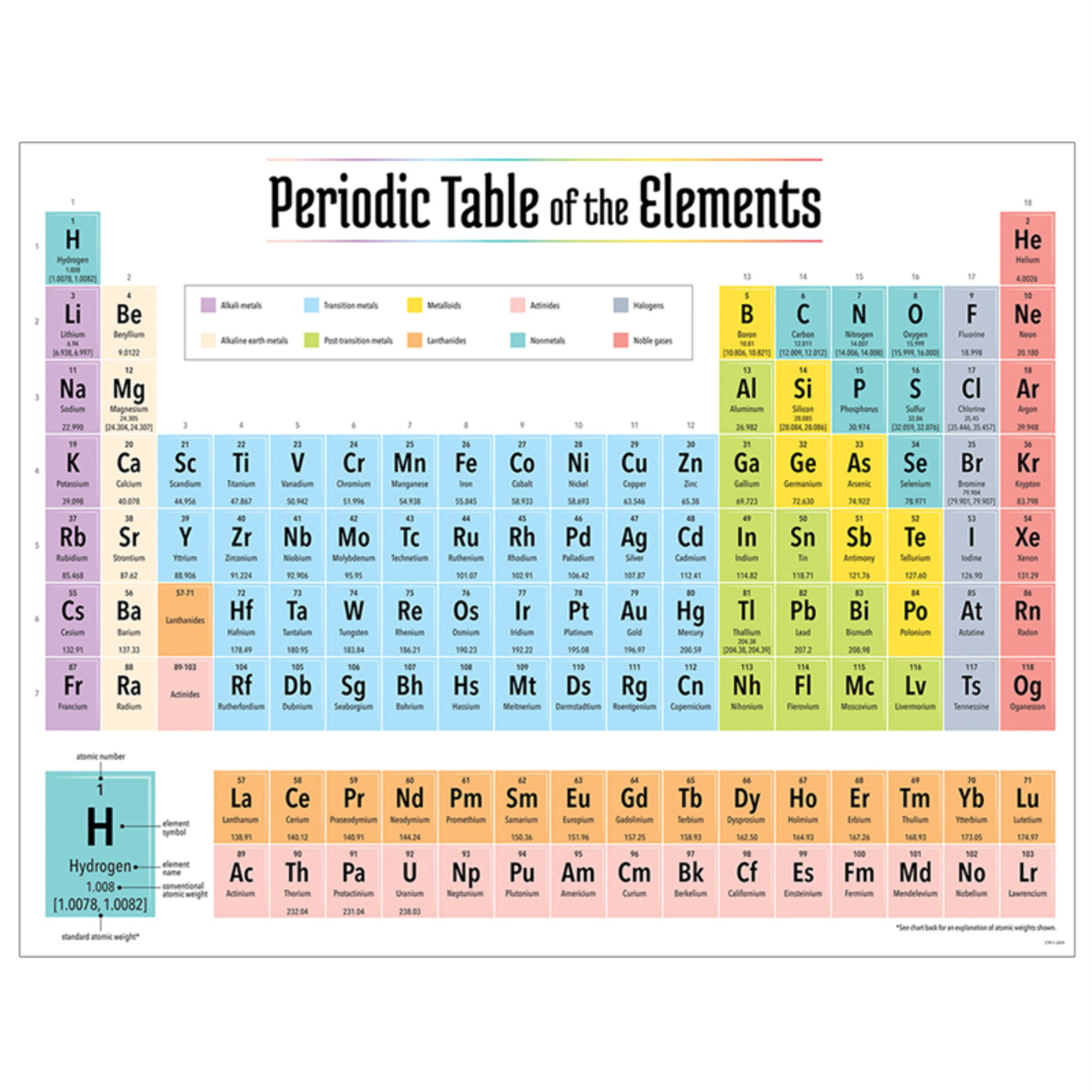
Each next element in a period has one more proton and is less metallic than its predecessor. Not all elements form stable negative ions in which case the electron affinity is zero or even positive. So the more negative the electron affinity the more favourable the electron addition process is. All elements in a row have the same number of electron shells. The electron affinity is the potential energy change of the atom when an electron is added to a neutral gaseous atom to form a negative ion. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. A period on the periodic table is a row of chemical elements. This group contains very reactive nonmetal elements. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. 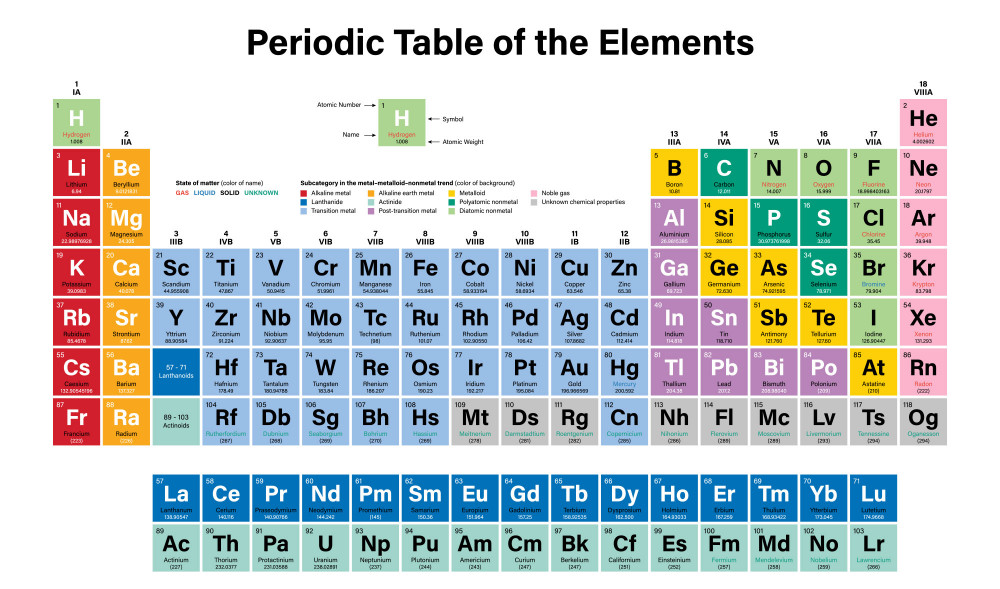
Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Group 1A is also known as the alkali metals. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
